Avian Influenza Inactivation
Fabrics that have undergone Cleanty Processing are expected to have antibacterial/antiviral effects and avian influenza inactivation. We can process cotton material with 99.9% suppression even after 50 washing tests. This processing technology is aimed at gauze mask textiles. Please note that it is patented and requires a contract for usage. Please contact us if you wish to have your fabric processed.
Patent No. 4832573: Antiviral and Antibacterial Processing Methods for Textile Structure
(Registration Date: September 30, 2011)
The Kanehisa Corporation assumes that Cleanty Processing may inactivate viral bacteria, based on their research and evaluation of antibacterial/MRSA measures.
The Kanehisa Corporation approached the Kyoto Prefectural University of Medicine in response to an avian influenza outbreak in Kyoto, requesting an avian influenza virus inactivation test for textiles utilizing Cleanty Processing technology. The said university cooperated with the tests conducted to verify antiviral effects against the virus (H5N2).
In February 2010, antiviral effects were confirmed for cotton textiles. (Confirmed decrease in 5 minutes)
Cotton: 90% or more reduction with Cleanty Processing only, 500 rounds friction test, and 50 times washing.
Polyester/Cotton: At least 90% reduction with Cleanty Processing only. At least 80% with 50 times washing
Silk, Rayon, Polyester: At least 90% reduction with Cleanty Processing only. Unconfirmed with washing test.
Functionality
| ① Antibacterial/Antiviral (at least 99% antibacterial rate) | [Testing Institution] |
|---|---|
| Avian Influenza Virus | [Kyoto Prefectural University of Medicine] |
| MRSA (Methicillin-resistant Staphylococcus aureus) | [Japan Chemical Fibers Association] |
| Staphylococcus aureus | [Kaken Test Center] |
| Klebsiella pneumoniae | [Kaken Test Center] |
| ② Deodorant (Ammonia/acetic acid gas removal 77%) | [Kaken Test Center] |
| ③ Antioxidant effects, yellowing prevention | |
| ④ Ultraviolet Protection Factor (96.4%) |
Cleanty Processing TechnologySilk Technology
Friendly to people and the environment
People are getting more and more conscious of protecting the environment and living pleasant and healthy lives. Cleanty Processing is a technology that imparts and boosts antiviral and bacteriostatic performance without using any chemicals that may harm the environment or your health. This is achieved through covalently binding molecules using safe agents and auxiliaries.
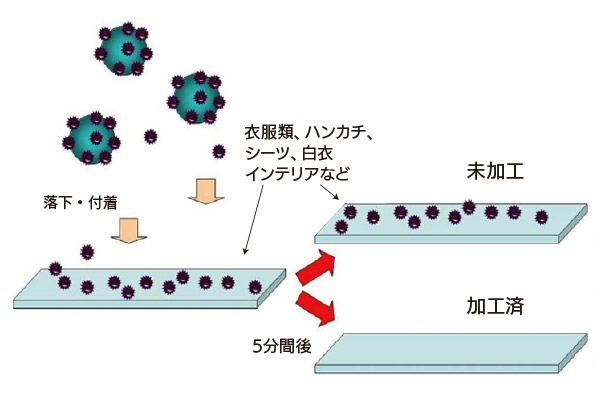
Antiviral Efficacy Test Results
Five-minute exposure to avian influenza H5N2 Virus reduction confirmed for the following five materials:
| Antiviral | |||
|---|---|---|---|
| (Five-minute exposure to avian influenza H5N2) | |||
| Material | Cleanty Processing only | 50 times washing | 500 rounds friction test |
| Cotton | -99.95% | -99.90% | -99.73% |
| Silk | -90.70% | Unconfirmed | Unconfirmed |
| Rayon | -98.69% | Unconfirmed | Unconfirmed |
| Polyester | -99.22% | Unconfirmed | Unconfirmed |
| Cotton/Polyester | -99.95% | -80.14% | Unconfirmed |
*Testing Institute: Kyoto Prefectural University of Medicine
INFORMATION
| Processing Technology | Cleanty Processing |
|---|---|
| Supported Materials | Cotton, polyester/cotton, silk/rayon/polyester |
| Applications | General clothing: casual clothing, uniforms, work clothes, work caps Bedding: Pillows, futons, sheets Medical: Masks, nursing care products, doctor coats, nurse coats Underwear: shorts, lingerie Industrial Materials: curtains, wallpapers, clothes, car seats, carpets, cloth bags (eco-bags) |
| Supported Width | 60〜120cm |
| Minimum Lot | Total of 200m (i.e. 50m x 4) |
| Estimated Delivery | approx. 10〜20 days |
Transaction Flow
Cleanty Processing uses patented chemicals and requires a patent license agreement to be availed of. The transaction flow is as follows:
- ① Please contact the person in charge of our processing plant (contact details below)
- ② The person in charge of our processing plant will contact Kanehisa Co., Ltd. (the patentee). Kanehisa Co., Ltd. will then contact you.
- ③ We'll be able to apply Cleanty Processing once your negotiations between Kanehisa Co., Ltd. are completed.
Inquiries
Operating Body: Tango Textile Industrial Association
3188 Koube, Omiya-cho, Kyotango-shi, Kyoto 629-2502
In-charge: Masaki Yamashita